Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
Initialising ...
 neutron diffraction
neutron diffractionKobayashi, Hiroki*; Komatsu, Kazuki*; Ito, Hayate*; Machida, Shinichi*; Hattori, Takanori; Kagi, Hiroyuki*
Journal of Physical Chemistry Letters (Internet), 14(47), p.10664 - 10669, 2023/11
Times Cited Count:1 Percentile:0.01(Chemistry, Physical)Ice IV is a metastable high-pressure phase of ice in which the water molecules exhibit orientational disorder. Although orientational ordering is commonly observed for other ice phases, it has not been reported for ice IV. We conducted  powder neutron diffraction experiments for DCl-doped D
powder neutron diffraction experiments for DCl-doped D O ice IV to investigate hydrogen ordering in ice IV. We found abrupt changes in the temperature derivative of unit cell volume, dV/dT, at about 120 K, and revealed their slightly ordered structure at low temperatures based on the Rietveld method. The occupancy of the D1 site deviates from 0.5; it increased when samples were cooled at higher pressures and reached 0.282(5) at 2.38 GPa, 58 K. Our results evidence the presence of a low-symmetry hydrogen-ordered state corresponding to ice IV. It seems, however, difficult to experimentally access the completely ordered phase corresponding to ice IV by slow cooling at high pressure.
O ice IV to investigate hydrogen ordering in ice IV. We found abrupt changes in the temperature derivative of unit cell volume, dV/dT, at about 120 K, and revealed their slightly ordered structure at low temperatures based on the Rietveld method. The occupancy of the D1 site deviates from 0.5; it increased when samples were cooled at higher pressures and reached 0.282(5) at 2.38 GPa, 58 K. Our results evidence the presence of a low-symmetry hydrogen-ordered state corresponding to ice IV. It seems, however, difficult to experimentally access the completely ordered phase corresponding to ice IV by slow cooling at high pressure.
Chiba, Kaori*; Matsui, Takuro*; Chatake, Toshiyuki*; Ohara, Takashi; Tanaka, Ichiro*; Yutani, Katsuhide*; Niimura, Nobuo*
Protein Science, 32(10), p.e4765_1 - e4765_13, 2023/10
Times Cited Count:0 Percentile:0(Biochemistry & Molecular Biology)Terasawa, Yukana*; Ohara, Takashi; Sato, Sota*; Yoshida, Satoshi*; Asahi, Toru*
Acta Crystallographica Section E; Crystallographic Communications (Internet), 78(3), p.306 - 312, 2022/03
Yano, Yoshio*; Ono, Toshikazu*; Ohara, Takashi; Hisaeda, Yoshio*
Chemistry; A European Journal, 27(71), p.17802 - 17807, 2021/12
Times Cited Count:4 Percentile:29.59(Chemistry, Multidisciplinary)Kwon, H.*; Pietrasiak, E.*; Ohara, Takashi; Nakao, Akiko*; Chae, B.*; Hwang, C.-C.*; Jung, D.*; Hwang, I.-C.*; Ko, Y. H.*; Kim, K.*; et al.
Inorganic Chemistry, 60(9), p.6403 - 6409, 2021/05
Times Cited Count:0 Percentile:0.01(Chemistry, Inorganic & Nuclear)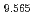 (Si
(Si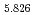 []
[]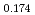 )O
)O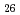 apatite without interstitial oxygens due to the overbonded channel oxygens
apatite without interstitial oxygens due to the overbonded channel oxygensFujii, Kotaro*; Yashima, Masatomo*; Hibino, Keisuke*; Shiraiwa, Masahiro*; Fukuda, Koichiro*; Nakayama, Susumu*; Ishizawa, Nobuo*; Hanashima, Takayasu*; Ohara, Takashi
Journal of Materials Chemistry A, 6(23), p.10835 - 10846, 2018/06
Times Cited Count:27 Percentile:69.76(Chemistry, Physical)Igarashi, Masayasu*; Matsumoto, Tomohiro*; Yagihashi, Fujio*; Yamashita, Hiroshi*; Ohara, Takashi; Hanashima, Takayasu*; Nakao, Akiko*; Moyoshi, Taketo*; Sato, Kazuhiko*; Shimada, Shigeru*
Nature Communications (Internet), 8, p.140_1 - 140_8, 2017/07
Times Cited Count:25 Percentile:63.68(Multidisciplinary Sciences)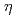
 -semiquinone complex; A Possible dielectric response mechanism
-semiquinone complex; A Possible dielectric response mechanismMitsumi, Minoru*; Ezaki, Kazunari*; Komatsu, Yuki*; Toriumi, Koshiro*; Miyato, Tatsuya*; Mizuno, Motohiro*; Azuma, Nobuaki*; Miyazaki, Yuji*; Nakano, Motohiro*; Kitagawa, Yasutaka*; et al.
Chemistry; A European Journal, 21(27), p.9682 - 9696, 2015/06
Times Cited Count:7 Percentile:24.93(Chemistry, Multidisciplinary)Ohara, Takashi
Nihon Kessho Gakkai-Shi, 56(5), p.301 - 306, 2014/10
no abstracts in English
 H(SeO
H(SeO )
)
Kiyanagi, Ryoji; Matsuo, Yasumitsu*; Ishikawa, Yoshihisa*; Noda, Yukio*; Ohara, Takashi; Kawasaki, Takuro; Oikawa, Kenichi; Kaneko, Koji; Tamura, Itaru; Hanashima, Takayasu*; et al.
no journal, ,
The materials represented as M H(XO
H(XO )
) (M=alkali metals, X=Se, S) are known to exhibit high protonic conductivities at relatively low temperature. Although the high protonic conductivity is considered to be due to the disorder of the hydrogen bonds in the materials, details have not been well understood. In addition, clarification of the phase transition to the high proton conducting phase is important in order to understand the realization of the high protonic conductivity. In this study, a high temperature neutron structural study was carried out on Rb
(M=alkali metals, X=Se, S) are known to exhibit high protonic conductivities at relatively low temperature. Although the high protonic conductivity is considered to be due to the disorder of the hydrogen bonds in the materials, details have not been well understood. In addition, clarification of the phase transition to the high proton conducting phase is important in order to understand the realization of the high protonic conductivity. In this study, a high temperature neutron structural study was carried out on Rb H(SeO
H(SeO )
) , and the solid solution of Rb
, and the solid solution of Rb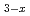 K
K H(SeO
H(SeO )
) were also investigated by means of conductivity measurements and neutron structural studies. The high temperature neutron structure analyses revealed that the protons in the high proton conducting phase were two dimensionally distributed, which is considered to be the direct observation of the conducting protons. The solid solution exhibited non-linear decreases of the phase transition temperatures as the K concentration increased. The K ions were found to prefer to occupy one of two possible sites, of which occupancies apparently have close relationship with the phase transition temperature.
were also investigated by means of conductivity measurements and neutron structural studies. The high temperature neutron structure analyses revealed that the protons in the high proton conducting phase were two dimensionally distributed, which is considered to be the direct observation of the conducting protons. The solid solution exhibited non-linear decreases of the phase transition temperatures as the K concentration increased. The K ions were found to prefer to occupy one of two possible sites, of which occupancies apparently have close relationship with the phase transition temperature.
Ohara, Takashi
no journal, ,
no abstracts in English
Ohara, Takashi; Kiyanagi, Ryoji; Hanashima, Takayasu*; Moyoshi, Taketo*; Nakao, Akiko*; Munakata, Koji*; Kuroda, Tetsuya*
no journal, ,
no abstracts in English
Ohara, Takashi; Kiyanagi, Ryoji; Oikawa, Kenichi; Kawasaki, Takuro; Kaneko, Koji; Tamura, Itaru; Nakao, Akiko*; Hanashima, Takayasu*; Munakata, Koji*
no journal, ,
Ohara, Takashi; Hanashima, Takayasu*; Munakata, Koji*; Moyoshi, Taketo*; Kiyanagi, Ryoji; Nakao, Akiko*; Kuroda, Tetsuya*
no journal, ,
no abstracts in English
Ohara, Takashi
no journal, ,
no abstracts in English
Ohara, Takashi
no journal, ,
no abstracts in English